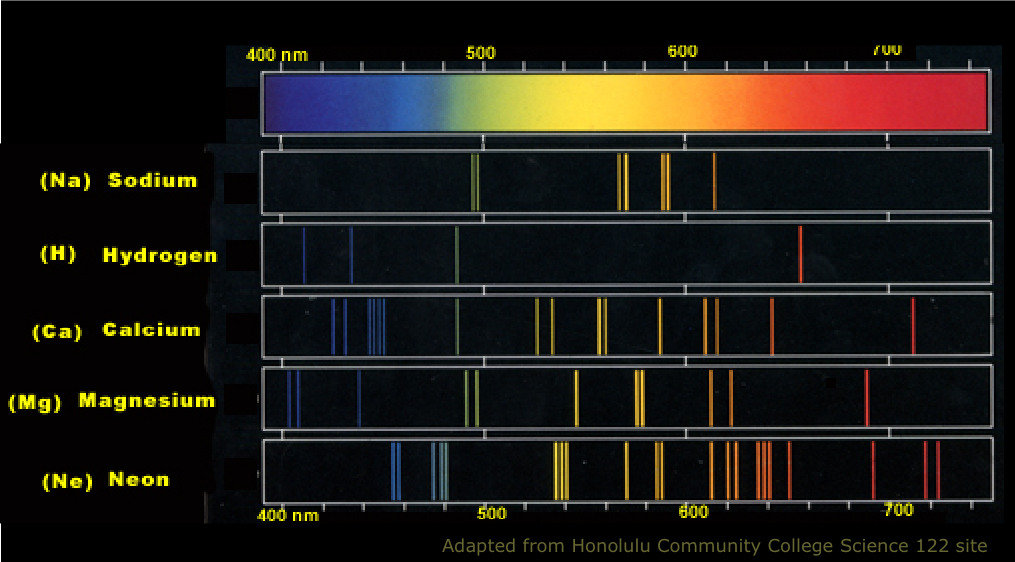
The complexes were irreversibly oxidized and reduced electrochemically and were weakly emissive at low concentrations (Stokes shifts between 23 and 31 nm), showing little solvent dependence. The resulting differences in the column densities (log N). The absorption spectra of the dimers were red-shifted relative to the parent β-diketimine ligands. These offsets correspond to 40 per cent of the inverse of the S/N of the spectrum near the absorption. The stability of the resulting dimeric structures relative to the corresponding monomeric, tetradentate boron complexes was studied computationally, and theory confirmed that the dimeric structures were strongly favored. To evaluate its chemical stability, the change in its UV-vis absorption spectrum in a solution of oxygen-saturated toluene in the dark was monitored. Calculated B K-edge x-ray absorption spectra (XAS) for different B. If the light is passed through a prism or diffraction grating, it is split into its various colors. Hydrogen absorption and emission lines in the visible spectrum. If a high voltage (5000 volts) is applied, the tube lights up with a bright pink glow. If boron emits radiation at 518 nm, what color will boron impart to a flame. These are the series which are in a sequence of lines corresponding to atomic transitions. A hydrogen discharge tube is a slim tube containing hydrogen gas at low pressure with an electrode at each end. The procedure of hydrogen absorption and emission give rise to series, each starting and ending with the same hydrogen atomic state. The resulting complexes were found to contain a unique hydrogen-bond-supported boron–oxygen–boron bridge, as confirmed by X-ray crystallography. These results suggest possible existence of BH complexes and/or B pairs in B-doped diamond. The hydrogen Spectrum is significant since the bulk portion of the Universe is made of hydrogen. The title complexes were isolated via demethylation of β-diketimine ligands and boron difluoride complexes bearing 2-anisole N-aryl substituents using boron tribromide. The synthetic routes employed were designed to circumvent the undesirable formation of β-ketimines and 2-methylbenzoxazoles observed when traditional synthetic routes toward the target β-diketiminate ligands were attempted. The infra-red absorption spectrum of silicon doped with high densities of boron and phosphorus has been measured from 1 to 60 m and at temperatures between. Two dimeric boron complexes of potentially tetradentate and trianionic β-diketiminate ligands bearing phenol substituents were prepared and characterized.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
